

It seemed too good to be true. Vaccines usually take years to develop. But in just months, multiple organizations announced development of vaccines against COVID-19.
What some people didn’t realize was that fast development was made possible by more than 10 years of vaccine research by several laboratories worldwide, including at Texas Children’s Center for Vaccine Development.
“Coronaviruses are threats that seem to arise out of nowhere and cause explosive epidemics and pandemics,” said Peter J. Hotez, MD, PhD, co-director of Texas Children’s Center for Vaccine Development and dean of the National School of Tropical Medicine at Baylor College of Medicine.
In 2002, SARS (severe acute respiratory syndrome) arose in South China and quickly spread internationally. In 2012, MERS (Middle East respiratory syndrome) began an outbreak on the Arabian Peninsula. Both were caused by coronaviruses, a large family of viruses that can cause the common cold to deadly diseases.
In December 2019, China reported a cluster of “viral pneumonia of unknown cause,” which was later identified as a novel coronavirus. By May 2021, deaths from COVID-19 totaled more than 594,000 people in the U.S. and more than 3.52 million people worldwide.
Simply put, the world did not have the luxury of waiting the normal development time for new vaccines. Luckily, we didn’t have to.
“We already had 10 years of experience in the coronavirus vaccine development space, and 20 years of experience in developing prototype vaccines,” said Maria Elena Bottazzi, PhD, co-director of Texas Children's Center for Vaccine Development and associate dean of the National School of Tropical Medicine. “The feeling of urgency that we and all our scientists had was of amazing intensity and stress. But at the same time, it was also quite rewarding because all our teams jumped in without hesitancy, recognizing it was our duty to respond rapidly.”
Dr. Hotez and Dr. Bottazzi have worked together for 20 years developing low-cost vaccines for diseases that primarily affect poverty-stricken populations around the world. These diseases are generally overlooked by large pharmaceutical companies because they have little chance for any intervention to be profitable.
About 10 years ago, a coronavirus vaccine research consortium was established by Texas Children’s Center for Vaccine Development, initially with scientists from the New York Blood Center and the Galveston National Lab at the University of Texas Medical Branch.
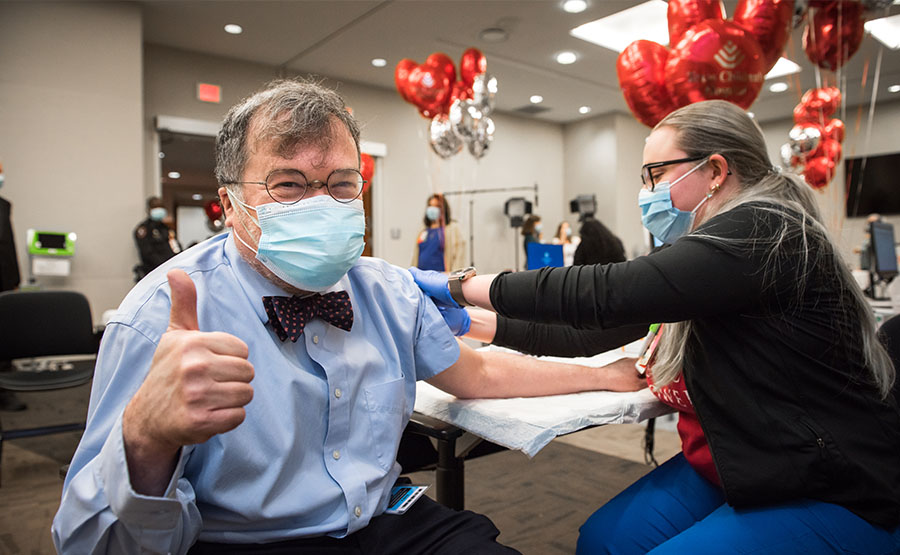 Peter J. Hotez, MD, PhD, receives the Pfizer COVID-19 vaccine. He co-leads development of another vaccine that’s low-cost and easy-to-use.
Peter J. Hotez, MD, PhD, receives the Pfizer COVID-19 vaccine. He co-leads development of another vaccine that’s low-cost and easy-to-use.
“We embraced this coronavirus vaccine program, and we've learned a lot over the last decade. We learned that the spike protein is the soft underbelly of the virus. We showed that if you deliver the receptor binding domain of the spike protein as a vaccine, it's highly effective. It induces what are called virus neutralizing antibodies,” Dr. Hotez said. The receptor binding domain is the part of the spike protein that allows it to dock to receptors so that the protein can enter cells and cause infection.
“One of the difficult features of this coronavirus is that it has two faces. On one hand, it can cause terrible cardiovascular and neurologic disease in one group of people. Then in another group of people, it is without any symptoms at all and has the ability to replicate in large amounts in our nose and mouth. That’s what made this such a difficult virus to control,” he said.
“All that work we did over the last decade helped lay the groundwork for the current generation of COVID-19 vaccines,” Dr. Hotez said.
Dr. Hotez emphasized that vaccines fast-tracked under U.S. Operation Warp Speed and programs in other countries still underwent rigorous testing for safety and effectiveness. With billions of dollars invested by governments and philanthropists, pharmaceutical companies were able to save time by conducting preclinical and multiple clinical trials simultaneously instead of sequentially.
The vaccine center at Texas Children’s and Baylor developed a low-cost COVID-19 vaccine that will be produced and evaluated by Biological E in India, one of the world’s largest vaccine producers. Biological E already reached major milestones. First, Biological E scaled-up the production to 1.2 billion doses for the recombinant protein vaccine antigen. Second, they completed a combined Phase 1-2 clinical trial in seven sites in India. As of May 2021, Biological E received approval to advance into Phase 3 clinical trials and ramp up production for the doses that would enable the vaccine to be authorized and deployed in low- and middle-income countries.
“If all we do is vaccinate the American people and don't do anything about the world's low- and middle-income countries, that creates a huge virus incubator for new variants to emerge. And that makes our nation susceptible as well. We have to figure out a way to vaccinate both the United States and the rest of the world. Otherwise, it's going to be self-defeating,” Dr. Hotez said.
“The problem that we face globally with these mRNA vaccines, like those from Pfizer and Moderna, is that the technology is too new to know how to scale it for the billions of doses that are needed, and you have this onerous cold-chain storage and transport requirement for uninterrupted extremely low temperature from production to distribution,” Dr. Hotez said.
“Our vaccine builds on the track record of another well-known vaccine. It's inexpensive – we think our vaccine could be produced for approximately $1.50 a dose. It only requires standard refrigeration. Ours is what's needed in Africa and Latin America, which is a durable, low-cost, easy-to-use vaccine,” he said.
The Baylor-Texas Children’s COVID-19 vaccine uses the same technology as the Hepatitis B vaccine, which has inoculated adults and children safely for 40 years.
“It's very likely that vaccines such as ours are also going to be ideal for use in pediatric populations because they are produced with technologies that are already used for other pediatric vaccines,” Dr. Bottazzi said.
Another mission of the Center for Vaccine Development is to strengthen and build capacity in other countries. The center’s scientists work with researchers in many regions, especially in Southeast Asia, the Middle East and Latin America, to help them bolster their own capabilities and know-how to produce vaccines for their own populations.
“The vaccine center serves as a unique model. Its approach, embedded in an academic health-hospital system, allows us to advance robust vaccine science, create vaccine prototypes and then work with large, multinational organizations to transfer the know-how for large-scale production,” Dr. Bottazzi said.
Dr. Hotez and Dr. Bottazzi credit Texas Children’s with enabling the center to make such an important contribution to combating COVID-19.
“In 2016, the priority of coronavirus research pretty much disappeared, and we were not able to obtain outside funding to continue the coronavirus vaccine program initiated in 2011. What allowed us to keep the program alive was our funding from Texas Children’s,” Dr. Bottazzi said.
“We continued learning about these vaccines; we kept studying them under strict quality control; we continued having our people trained and intellectually on top of coronavirus scientific advancements. That’s the point of Texas Children’s support for our vaccine center – they are so forward-thinking that we’re allowed to invest into cutting-edge programs even though others may not feel the urgency,” she said.
The decade of experience in the field made possible the additional role of explaining to the U.S. and the world what COVID-19 is and what individuals can do to protect themselves and their families. In 2020, Dr. Hotez gave 683 television and radio interviews about COVID-19, published 28 articles and two books, and wrote 10 op-eds. Dr. Bottazzi, in turn, took on sharing knowledge in Latin America and the rest of the world, explaining how vaccines are developed and why vaccines are so important, especially for underserved populations. Her multilingual vaccine engagement reached a potential audience of more than 15 billion via blogs, TV appearances and online news outlets.
“We were among the few that had been working on coronavirus research for the last 10 years, and that brought with it enormous credibility,” Dr. Hotez said. “We were able to speak authoritatively on why it was necessary to wear masks and do social distancing and why COVID-19 vaccines were necessary. We were involved at every level in terms of vaccine development, other biotherapeutic measures and shaping policy for coronaviruses.”
For the vaccine center, 2020 was a year of extraordinary intensity and fulfillment, strengthening capacity, teaching, training, shaping policy and creating vaccines.
For more information about COVID-19 vaccines, please see articles co-authored by Dr. Hotez and Dr. Bottazzi, among others: